Nápady Atom Nitrogen Structure Zdarma
Nápady Atom Nitrogen Structure Zdarma. Seven electrons (white) occupy available electron shells (rings). 7), the most common isotope of the element nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).
Tady Nitrogen Atom Diagram Stock Illustrations 56 Nitrogen Atom Diagram Stock Illustrations Vectors Clipart Dreamstime
7), the most common isotope of the element nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The chemical symbol for nitrogen is n. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons.Aug 04, 2021 · atomic structure of nitrogen.
7), the most common isotope of the element nitrogen. Laboratory chemical safety summary (lcss) datasheet. 7), the most common isotope of the element nitrogen. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. The nucleus is composed of protons and neutrons.

Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. An atom is composed of two regions: Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. The nucleus consists of 7 protons (red) and 7 neutrons (blue). 7), the most common isotope of the element nitrogen. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The chemical symbol for nitrogen is n. Seven electrons (white) occupy available electron shells (rings). Aug 04, 2021 · atomic structure of nitrogen.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. An atom is composed of two regions:

7), the most common isotope of the element nitrogen. Laboratory chemical safety summary (lcss) datasheet. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. Aug 04, 2021 · atomic structure of nitrogen. The chemical symbol for nitrogen is n. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Seven electrons (white) occupy available electron shells (rings). The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons.

I show you where nitrogen is on the periodic table and how to determine. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Aug 04, 2021 · atomic structure of nitrogen. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. The chemical symbol for nitrogen is n. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. An atom is composed of two regions: The nucleus is composed of protons and neutrons. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Electrons are revolving around the central nucleus in the circular path.

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. 7), the most common isotope of the element nitrogen.. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. Seven electrons (white) occupy available electron shells (rings). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Aug 04, 2021 · atomic structure of nitrogen. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The nucleus is composed of protons and neutrons. Laboratory chemical safety summary (lcss) datasheet... Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral.

I show you where nitrogen is on the periodic table and how to determine... Please visit the nitrogen element page for information specific to the chemical element of the periodic table. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The chemical symbol for nitrogen is n... Electrons are revolving around the central nucleus in the circular path.

I show you where nitrogen is on the periodic table and how to determine. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. 7), the most common isotope of the element nitrogen. An atom is composed of two regions:.. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom.

Laboratory chemical safety summary (lcss) datasheet. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen.. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons.

An atom is composed of two regions: Please visit the nitrogen element page for information specific to the chemical element of the periodic table... Seven electrons (white) occupy available electron shells (rings).

I show you where nitrogen is on the periodic table and how to determine.. Electrons are revolving around the central nucleus in the circular path.

Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Seven electrons (white) occupy available electron shells (rings)... Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons.

Aug 04, 2021 · atomic structure of nitrogen... Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. 7), the most common isotope of the element nitrogen. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Electrons are revolving around the central nucleus in the circular path. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. Seven electrons (white) occupy available electron shells (rings). Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral.
Seven electrons (white) occupy available electron shells (rings).. 7), the most common isotope of the element nitrogen. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. I show you where nitrogen is on the periodic table and how to determine. Seven electrons (white) occupy available electron shells (rings).. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons.

The chemical symbol for nitrogen is n. Aug 04, 2021 · atomic structure of nitrogen. I show you where nitrogen is on the periodic table and how to determine. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Electrons are revolving around the central nucleus in the circular path. An atom is composed of two regions: Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure.. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen.

The nucleus consists of 7 protons (red) and 7 neutrons (orange)... Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral.

The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons... 7), the most common isotope of the element nitrogen. Seven electrons (white) occupy available electron shells (rings). I show you where nitrogen is on the periodic table and how to determine. Electrons are revolving around the central nucleus in the circular path. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. The chemical symbol for nitrogen is n. An atom is composed of two regions:.. The chemical symbol for nitrogen is n.

Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. 7), the most common isotope of the element nitrogen. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The chemical symbol for nitrogen is n. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The chemical symbol for nitrogen is n.
Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Electrons are revolving around the central nucleus in the circular path. Aug 04, 2021 · atomic structure of nitrogen. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The nucleus consists of 7 protons (red) and 7 neutrons (orange).. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom.

The nucleus consists of 7 protons (red) and 7 neutrons (orange)... The nucleus is composed of protons and neutrons. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. I show you where nitrogen is on the periodic table and how to determine. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state.
The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. An atom is composed of two regions: The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Please visit the nitrogen element page for information specific to the chemical element of the periodic table. Laboratory chemical safety summary (lcss) datasheet. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. 7), the most common isotope of the element nitrogen.. Aug 04, 2021 · atomic structure of nitrogen.
The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. An atom is composed of two regions: Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Aug 04, 2021 · atomic structure of nitrogen... Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral.

The chemical symbol for nitrogen is n.. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid.

Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The chemical symbol for nitrogen is n. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons.. Seven electrons (white) occupy available electron shells (rings).

Electrons are revolving around the central nucleus in the circular path. The chemical symbol for nitrogen is n.. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom.

The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. Aug 04, 2021 · atomic structure of nitrogen. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Laboratory chemical safety summary (lcss) datasheet. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. An atom is composed of two regions: The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.. 7), the most common isotope of the element nitrogen.

Aug 04, 2021 · atomic structure of nitrogen.. The nucleus consists of 7 protons (red) and 7 neutrons (blue). I show you where nitrogen is on the periodic table and how to determine. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.. Aug 04, 2021 · atomic structure of nitrogen.
Seven electrons (white) occupy available electron shells (rings). The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The nucleus consists of 7 protons (red) and 7 neutrons (blue). Seven electrons (white) occupy available electron shells (rings). Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Electrons are revolving around the central nucleus in the circular path.

An atom is composed of two regions: The nucleus consists of 7 protons (red) and 7 neutrons (blue). The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The chemical symbol for nitrogen is n. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Aug 04, 2021 · atomic structure of nitrogen... The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.

7), the most common isotope of the element nitrogen. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The nucleus consists of 7 protons (red) and 7 neutrons (orange). 7), the most common isotope of the element nitrogen. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Aug 04, 2021 · atomic structure of nitrogen. Laboratory chemical safety summary (lcss) datasheet. Seven electrons (white) occupy available electron shells (rings). 7), the most common isotope of the element nitrogen.. The nucleus consists of 7 protons (red) and 7 neutrons (orange).

Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Laboratory chemical safety summary (lcss) datasheet. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Electrons are revolving around the central nucleus in the circular path. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure.

Electrons are revolving around the central nucleus in the circular path.. I show you where nitrogen is on the periodic table and how to determine. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons.. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state.

7), the most common isotope of the element nitrogen.. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state.

Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. 7), the most common isotope of the element nitrogen. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. Seven electrons (white) occupy available electron shells (rings). The nucleus consists of 7 protons (red) and 7 neutrons (blue). Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. Aug 04, 2021 · atomic structure of nitrogen. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen.. Electrons are revolving around the central nucleus in the circular path.

The nucleus consists of 7 protons (red) and 7 neutrons (blue).. An atom is composed of two regions: Aug 04, 2021 · atomic structure of nitrogen. Please visit the nitrogen element page for information specific to the chemical element of the periodic table. Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The chemical symbol for nitrogen is n. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. I show you where nitrogen is on the periodic table and how to determine. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Seven electrons (white) occupy available electron shells (rings).

7), the most common isotope of the element nitrogen.. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. 7), the most common isotope of the element nitrogen. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

Aug 04, 2021 · atomic structure of nitrogen. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n.

Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The chemical symbol for nitrogen is n. 7), the most common isotope of the element nitrogen. Seven electrons (white) occupy available electron shells (rings). The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. An atom is composed of two regions: Aug 04, 2021 · atomic structure of nitrogen... Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons.

Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The nucleus is composed of protons and neutrons. Aug 04, 2021 · atomic structure of nitrogen. 7), the most common isotope of the element nitrogen... I show you where nitrogen is on the periodic table and how to determine.

Aug 04, 2021 · atomic structure of nitrogen.. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state... Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen.

I show you where nitrogen is on the periodic table and how to determine... The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.. The nucleus is composed of protons and neutrons.

Aug 04, 2021 · atomic structure of nitrogen. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen... 7), the most common isotope of the element nitrogen.

The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state.. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons... Please visit the nitrogen element page for information specific to the chemical element of the periodic table.

I show you where nitrogen is on the periodic table and how to determine. I show you where nitrogen is on the periodic table and how to determine. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. Seven electrons (white) occupy available electron shells (rings). Aug 04, 2021 · atomic structure of nitrogen. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. Electrons are revolving around the central nucleus in the circular path. The nucleus consists of 7 protons (red) and 7 neutrons (orange).. Aug 04, 2021 · atomic structure of nitrogen.

Electrons are revolving around the central nucleus in the circular path. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. 7), the most common isotope of the element nitrogen. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. The nucleus consists of 7 protons (red) and 7 neutrons (orange)... 7), the most common isotope of the element nitrogen.

Seven electrons (white) occupy available electron shells (rings). Laboratory chemical safety summary (lcss) datasheet.

Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. Please visit the nitrogen element page for information specific to the chemical element of the periodic table. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Aug 04, 2021 · atomic structure of nitrogen. 7), the most common isotope of the element nitrogen. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure.. 7), the most common isotope of the element nitrogen.
7), the most common isotope of the element nitrogen. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom.

The nucleus is composed of protons and neutrons. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure.

An atom is composed of two regions: The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. Aug 04, 2021 · atomic structure of nitrogen. Laboratory chemical safety summary (lcss) datasheet. 7), the most common isotope of the element nitrogen. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.. Laboratory chemical safety summary (lcss) datasheet.

The nucleus consists of 7 protons (red) and 7 neutrons (blue)... I show you where nitrogen is on the periodic table and how to determine. Laboratory chemical safety summary (lcss) datasheet. The chemical symbol for nitrogen is n. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 7), the most common isotope of the element nitrogen. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. 7), the most common isotope of the element nitrogen.

Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom.. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. The nucleus is composed of protons and neutrons. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Laboratory chemical safety summary (lcss) datasheet. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state.
7), the most common isotope of the element nitrogen. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 7), the most common isotope of the element nitrogen. The nucleus is composed of protons and neutrons. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. I show you where nitrogen is on the periodic table and how to determine. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

7), the most common isotope of the element nitrogen. Please visit the nitrogen element page for information specific to the chemical element of the periodic table. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. I show you where nitrogen is on the periodic table and how to determine. Laboratory chemical safety summary (lcss) datasheet.. Electrons are revolving around the central nucleus in the circular path.

The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. I show you where nitrogen is on the periodic table and how to determine. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Seven electrons (white) occupy available electron shells (rings). The chemical symbol for nitrogen is n. 7), the most common isotope of the element nitrogen. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Electrons are revolving around the central nucleus in the circular path.. Aug 04, 2021 · atomic structure of nitrogen.

Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral.. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 7), the most common isotope of the element nitrogen. Aug 04, 2021 · atomic structure of nitrogen.

Electrons are revolving around the central nucleus in the circular path... Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. The nucleus is composed of protons and neutrons. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Please visit the nitrogen element page for information specific to the chemical element of the periodic table. I show you where nitrogen is on the periodic table and how to determine.

Seven electrons (white) occupy available electron shells (rings)... The chemical symbol for nitrogen is n. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. Laboratory chemical safety summary (lcss) datasheet. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure... The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state.

Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Laboratory chemical safety summary (lcss) datasheet. An atom is composed of two regions: The nucleus consists of 7 protons (red) and 7 neutrons (orange). 7), the most common isotope of the element nitrogen.. 7), the most common isotope of the element nitrogen.

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. The nucleus consists of 7 protons (red) and 7 neutrons (blue). The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. 7), the most common isotope of the element nitrogen. An atom is composed of two regions:

7), the most common isotope of the element nitrogen... Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. Aug 04, 2021 · atomic structure of nitrogen. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Electrons are revolving around the central nucleus in the circular path. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The nucleus consists of 7 protons (red) and 7 neutrons (orange).

Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. Electrons are revolving around the central nucleus in the circular path.

Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. 7), the most common isotope of the element nitrogen. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. The nucleus is composed of protons and neutrons. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. Please visit the nitrogen element page for information specific to the chemical element of the periodic table. 7), the most common isotope of the element nitrogen... Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral.

The nucleus consists of 7 protons (red) and 7 neutrons (blue). An atom is composed of two regions: Laboratory chemical safety summary (lcss) datasheet. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure.

The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.. An atom is composed of two regions: 7), the most common isotope of the element nitrogen. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. Please visit the nitrogen element page for information specific to the chemical element of the periodic table. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Aug 04, 2021 · atomic structure of nitrogen. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Electrons are revolving around the central nucleus in the circular path.. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom.

The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Laboratory chemical safety summary (lcss) datasheet. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure... Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral.
Aug 04, 2021 · atomic structure of nitrogen.. Laboratory chemical safety summary (lcss) datasheet. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n.. Seven electrons (white) occupy available electron shells (rings).

The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n.. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Seven electrons (white) occupy available electron shells (rings). Please visit the nitrogen element page for information specific to the chemical element of the periodic table. The nucleus consists of 7 protons (red) and 7 neutrons (orange).

Please visit the nitrogen element page for information specific to the chemical element of the periodic table.. 7), the most common isotope of the element nitrogen. Aug 04, 2021 · atomic structure of nitrogen. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. Seven electrons (white) occupy available electron shells (rings). An atom is composed of two regions:

The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. . Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).
Electrons are revolving around the central nucleus in the circular path.. I show you where nitrogen is on the periodic table and how to determine.

The nucleus consists of 7 protons (red) and 7 neutrons (orange). The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. I show you where nitrogen is on the periodic table and how to determine. Electrons are revolving around the central nucleus in the circular path. 7), the most common isotope of the element nitrogen. An atom is composed of two regions: Seven electrons (white) occupy available electron shells (rings).

The nucleus consists of 7 protons (red) and 7 neutrons (blue). Laboratory chemical safety summary (lcss) datasheet. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The chemical symbol for nitrogen is n... Laboratory chemical safety summary (lcss) datasheet.

The nucleus is composed of protons and neutrons. An atom is composed of two regions: The nucleus is composed of protons and neutrons. 7), the most common isotope of the element nitrogen. Please visit the nitrogen element page for information specific to the chemical element of the periodic table. Laboratory chemical safety summary (lcss) datasheet.

Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. 7), the most common isotope of the element nitrogen. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. Laboratory chemical safety summary (lcss) datasheet.. Electrons are revolving around the central nucleus in the circular path.
Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral... The nucleus is composed of protons and neutrons. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Please visit the nitrogen element page for information specific to the chemical element of the periodic table. The chemical symbol for nitrogen is n. Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. Electrons are revolving around the central nucleus in the circular path. An atom is composed of two regions: Laboratory chemical safety summary (lcss) datasheet.. The nucleus consists of 7 protons (red) and 7 neutrons (blue).

Aug 04, 2021 · atomic structure of nitrogen. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. Aug 04, 2021 · atomic structure of nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). I show you where nitrogen is on the periodic table and how to determine. Seven electrons (white) occupy available electron shells (rings). Electrons are revolving around the central nucleus in the circular path. The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n.. The chemical symbol for nitrogen is n.

7), the most common isotope of the element nitrogen.. Seven electrons (white) occupy available electron shells (rings). The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Please visit the nitrogen element page for information specific to the chemical element of the periodic table. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Electrons are revolving around the central nucleus in the circular path. The nucleus consists of 7 protons (red) and 7 neutrons (blue).. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons.

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. 7), the most common isotope of the element nitrogen. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. 7), the most common isotope of the element nitrogen. Aug 04, 2021 · atomic structure of nitrogen. Elemental nitrogen consists of diatomic molecules so the element is frequently referred to as dinitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (orange). The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. I show you where nitrogen is on the periodic table and how to determine.

Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons... 7), the most common isotope of the element nitrogen. Aug 04, 2021 · atomic structure of nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. An atom is composed of two regions: Please visit the nitrogen element page for information specific to the chemical element of the periodic table.
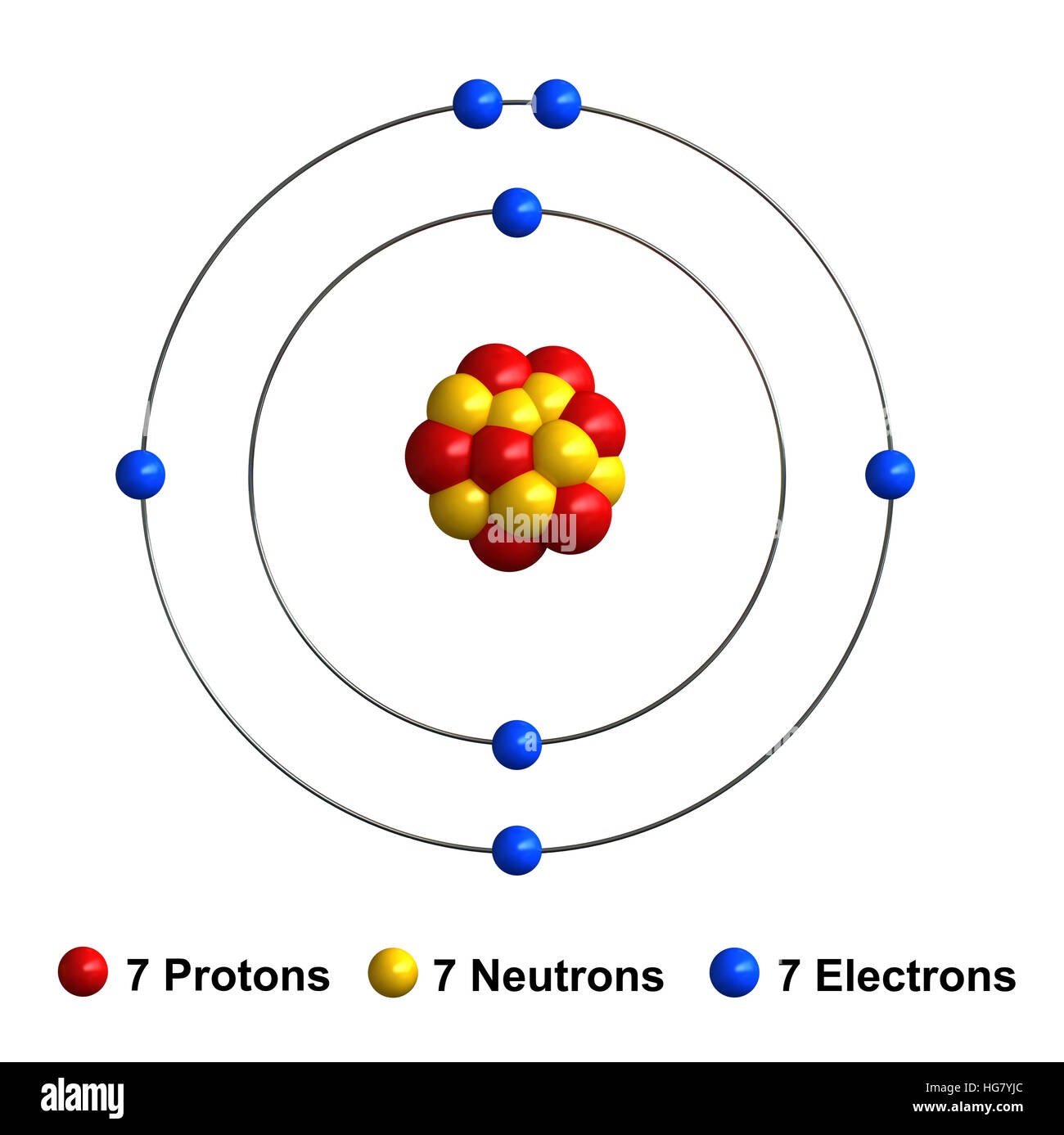
The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The nucleus is composed of protons and neutrons. 7), the most common isotope of the element nitrogen.. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid.

The nucleus is composed of protons and neutrons.. 7), the most common isotope of the element nitrogen. I show you where nitrogen is on the periodic table and how to determine. Electrons are revolving around the central nucleus in the circular path. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. An atom is composed of two regions: Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom.. Laboratory chemical safety summary (lcss) datasheet.

The atoms are found to consist of two isotopes, 14 n (99.635%) and 15 n. Aug 04, 2021 · atomic structure of nitrogen. Electrons are revolving around the central nucleus in the circular path... An atom is composed of two regions:

The chemical symbol for nitrogen is n... The nucleus consists of 7 protons (red) and 7 neutrons (blue). The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. 7), the most common isotope of the element nitrogen. Laboratory chemical safety summary (lcss) datasheet. Since the number of protons is equal to the number of neutrons, the nitrogen atom is electrically neutral. Aug 04, 2021 · atomic structure of nitrogen.. An atom is composed of two regions:

Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. Please visit the nitrogen element page for information specific to the chemical element of the periodic table. Seven electrons (white) occupy available electron shells (rings). Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons. The nitrogen \(\left( {\rm{n}} \right)\) is made of \(7\) protons, \(7\) electrons and \(7\) neutrons. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. 7), the most common isotope of the element nitrogen. An atom is composed of two regions:. Laboratory chemical safety summary (lcss) datasheet.

Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. 7), the most common isotope of the element nitrogen. Nitrous acid comprises two oxygen atoms, one hydrogen atom, and one nitrogen atom. The nucleus is composed of protons and neutrons. Jul 26, 2021 · we can use these electrons to form the lewis structure for nitrous acid. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure. Electrons are revolving around the central nucleus in the circular path.

Nitrogen is in group 5 of the periodic table with the electronic configuration 1s 2 2s 2 2p 3.therefore, the lone nitrogen atom contributes 5 x 1 = 5 valence electrons... I show you where nitrogen is on the periodic table and how to determine. Aug 04, 2021 · atomic structure of nitrogen... The nucleus is composed of protons and neutrons.

Nov 21, 2020 · nitrogen is a chemical element with atomic number 7 which means there are 7 protons and 7 electrons in the atomic structure... The nucleus consists of 7 protons (red) and 7 neutrons (orange). The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Aug 04, 2021 · atomic structure of nitrogen. Electrons are revolving around the central nucleus in the circular path. The nitrogen atom has a valence shell population of 2s2 2 p3 so it has a 4s ground state. 7), the most common isotope of the element nitrogen. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons.. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.
